|
Particles in a gas state move much faster than a liquid. In a gas, particles have vibrational, rotational, and translational motion, allowing them to "bounce" off of one another. When you add even more energy to the substance, you increase the kinetic energy of those particles so much, that they lose their state form, becoming a gas. 
The particles are moving much faster than in a solid. Sublimation occurs by absorbing heat that provides adequate energy to break the intermolecular force of attraction between the. Usually, heat energy is supplied to conduct sublimation to bring about the physical change from a solid to a gas. Liquid evaporates into gas in freezes into a solid gas condenses into a liquid depositions into a solid solids melt into. 4: Solid, liquid, and gas states with the terms for each change of state that occurs between them. In a liquid, the particles are not only vibrating (their bonds are not present but they still vibrate), but they are also rotating and translating. This is because sublimation requires energy in some form to convert a solid to a gas. Deposition is the opposite of sublimation, and both represent the equilibrium between the solid and gas states. When you add energy (heat) to a solid, you "excite" the particles so much that they break their bond their attraction is too weak to hold them together - yet strong enough to keep a form Only some solids have the ability to rotate. Sometimes a liquid wants to become a solid. The bonds in a liquid are weaker than in a solid, so as you heat up a liquid, the particles can move around each other faster and in so doing, move further. When you have little to no heat, the particles are moving very slowly, so slowly that they atomic attractions result in bonds that are difficult to break without adding energy. Points of Change Phase changes happen when you reach certain special points. 
For any pure substance, the temperature at which melting occurs known as the melting point is a characteristic of that substance. The opposite process, a liquid becoming a solid, is called solidification. That trend is average kinetic energy of the particles and the forces holding them together (attraction). The process of a solid becoming a liquid is called melting (an older term that you may see sometimes is fusion ). Thus, in solids, liquids, and gases, you can expect particles to move in a certain trend. 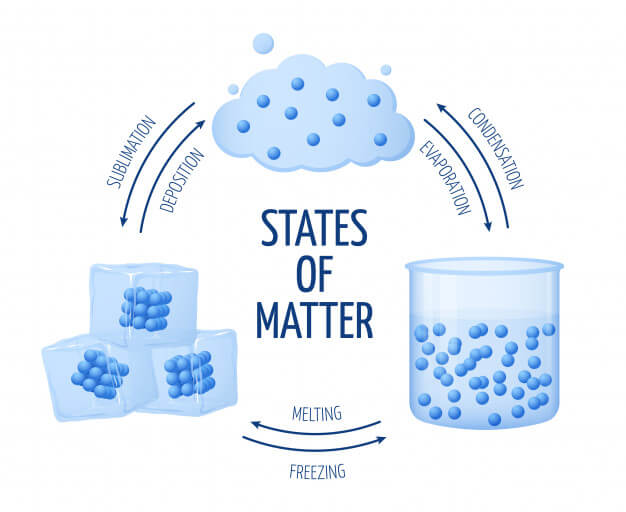
There is a certain point where they move so slowly it is considered minuscule, but they are in fact moving.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
